
TL;DR — Quick Summary
Carbon Dioxide Gas
Carbon dioxide is a colorless, odorless gas formed during respiration, combustion, and many chemical reactions. It is slightly soluble in water and heavier than air.
| Symbol | Molecular Weight |
| \(CO_2\) | 44 |
Laboratory Preparation of Carbon Dioxide Gas
When Calcium Carbonate is treated with dilute Hydrochloric Acid, Carbon Dioxide gas is produced.
Testing of Carbon Dioxide Gas
The produced carbon dioxide gas is not visible to us so we must have some tests to confirm it. Some tests are as follows:
- When a wet blue litmus paper is inserted into the gas jar containing carbon dioxide, it turns red color due to the formation of carbonic acid.
- When burning matchstick is placed near the mouth of gas jar containing carbon dioxide gas, it extinguishes because carbon dioxide gas Is non-supporter of combustion.
Properties of
Carbon Dioxide Gas
Physical Properties of Carbon Dioxide Gas
- Colorless and odorless gas
- 1.5 times Heavier than air
- Slightly soluble in water and forms Carbonic Acid \((H_2CO_3)\)
- At \(-78^0 C\) it turns into solid called dry ice.
- Non-combustible and does not support burning
- Can be liquefied under high pressure
Chemical Properties of Carbon Dioxide Gas
1. Magnesium Oxide is formed when burning magnesium ribbon is placed in test tube containing carbondioxide.
\(Mg + CO_2 \rightarrow MgO + C\)
2. Forms carbonic acid when dissolved in wate
\(CO_2 + H_2O \rightarrow H_2CO_3\)
3. Turns lime water milky due to formation of calcium carbonate
(When \(CO_2\) passed for a short time.)
\(Ca(OH)_2 + CO_2 \rightarrow CaCO_3 \downarrow + H_2O\)
(When \(CO_2\) is passed for a long time, the milky solution turns colorless.)
\(CaCO_3 + CO_2 + H_2O \rightarrow Ca(HCO_3)_2\)
4. Potassium Carbonate is formed when treated with Potassium Hydroxide.
\(KOH + CO_2 \rightarrow K_2CO_3 + H_2O\)
5. Plants make their food in the presence of sunlight and chlorophyll.
\(CO_2 + H_20 \xrightarrow[\text{chlorophyll}]{sunlight} C_6H_{12}O_6 + O_2\)
6. Carbon monoxide is formed when treated with red hot coke at \(900^0 C\)
\(CO_2 + C \rightarrow 2CO\)
7. Reacts with metal oxides to form carbonates
\(Na_2O + CO_2 \rightarrow Na_2CO_3\)
Uses of Carbon Dioxide Gas
- In fire extinguishers
- For making soda water and cold drinks
- In the photosynthesis process by plants
- As dry ice (solid CO₂) for refrigeration
- In greenhouses to promote plant growth
Ammonia Gas
Ammonia is a colorless gas with a sharp, pungent smell. It is highly soluble in water and forms ammonium hydroxide solution.
| Symbol | Molecular Weight |
| \(NH_3\) | 17 |
Laboratory Preparation of Ammonia Gas
When the mixture of Ammonium Chloride and Calcium Hydroxide is heated, Ammonia gas is produced.
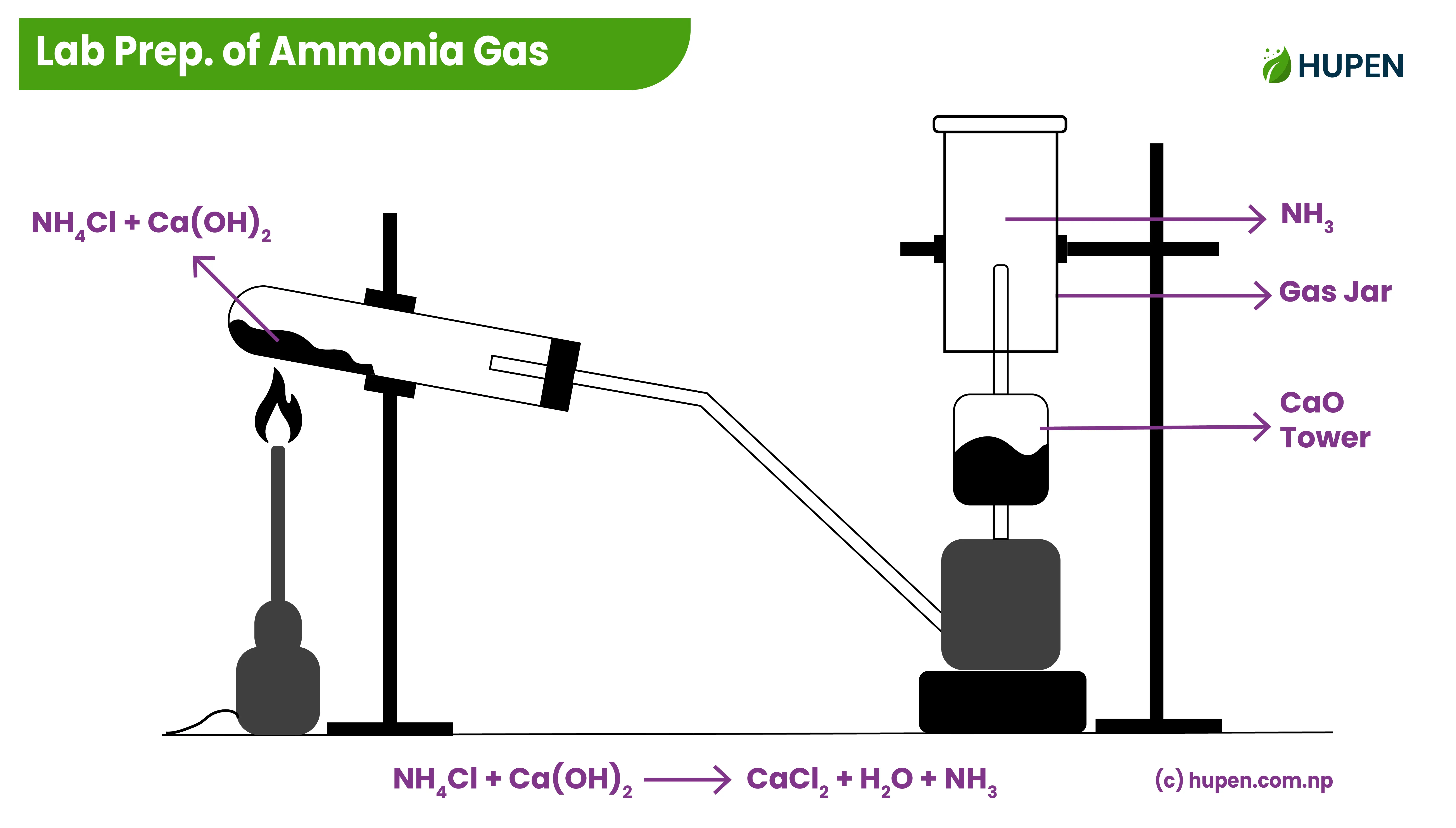
Testing of Ammonia Gas
The produced Ammonia gas is not visible to us so we must have some tests to confirm it. Some tests are as follows:
- When a wet red litmus paper is inserted into the gas jar containing ammonia, it turns blue color due to the formation of ammonium hydroxide base.
- The gas produces a white fume of ammonium chloride when reacted with concentrated hydrochloric acid.
Properties of Ammonia Gas
Physical Properties of Ammonia Gas
- Colorless gas with a sharp, irritating smell
- Lighter than air
- Highly soluble in water
- Forms a basic solution (ammonium hydroxide) in water
- Easily liquefied under pressure
Chemical Properties of Ammonia Gas
1. It is soluble in water to form Ammonium Hydroxide.
\( NH_3 + H_2O \rightarrow NH_4OH\)
2. Reacts with acids to form ammonium salts
\( NH_3 + HCl \rightarrow NH_4Cl \)
3. Ammonium Hydroxide reacts with acid to form salt and water.
\(NH_4OH + HCl \rightarrow NH_4Cl + H_2O\)
4. In the presence of high pressure and it reacts with carbon dioxide to form Urea.
\(NH_3 + CO_2 \xrightarrow[\text{Pressure}]{1500^0C} NH_2 – CO – NH_2 + H_2O\)
5. Reacts with oxygen on heating to form nitrogen and water
\(4NH_3 + 3O_2 \rightarrow 2N_2 + 6H_2O \)
6. When mixture of ammonia and oxygen at a temperature \(800^0 C\) is passed to the Platinum gauge, Nitric Oxide is formed.
\(4NH_3 + 5O_2 \xrightarrow[\text{Pt}]{800^0C} 6H_2O + 2NO\)
7. When ammonia gas is passed to the molten sodium metal, sodamide and hydrogen gas are formed.
\(2NH_3 + 2Na \rightarrow 2NaNH_2 + H_2\)
Uses of Ammonia Gas
- Manufacturing fertilizers (urea, ammonium nitrate)
- Used as a refrigerant in cold storage
- Production of nitric acid
- Used in cleaning agents
- In textile and pharmaceutical industries
Greenhouse Effect
The greenhouse effect is the natural process in which the Earth’s atmosphere traps some of the Sun’s heat to keep the planet warm enough to support life. Gases Like CO₂, CH₄, water vapor, and CFCs act as greenhouse gases.
Without the greenhouse effect, Earth would be too cold for living organisms.
Artificial Greenhouse
An artificial greenhouse is a structure made of glass or transparent materials where plants are grown under controlled temperature, humidity, and light.
Sunlight enters the structure, gets trapped inside, and keeps the greenhouse warm—similar to the natural greenhouse effect.
Importance and Utility of Artificial Greenhouse
- Helps grow plants in cold climates
- Protects plants from wind, frost, and pests
- Allows off-season cultivation
- Maintains constant temperature for better yield
- Used for research and commercial farming
Acid Rain
Acid rain is rainwater that becomes acidic due to the presence of sulfur dioxide (SO₂) and nitrogen oxides (NOₓ) released from industries, vehicles, and burning fossil fuels.
These gases react with water vapor in the atmosphere to form acids:
\(SO_2 + H_2O \rightarrow H_2SO_3\)
\(2NO_2 + H_2O \rightarrow HNO_2 + HNO_3\)
Effects of Acid Rain
- Damages crops, forests, and soil fertility
- Corrodes buildings, monuments, and metals
- Makes lakes and rivers acidic, harming aquatic life
- Causes skin and eye irritation in humans
Share Now
Share to help more learners!


Chapter Discussion
0 Comments
Log in to join the discussion
No comments yet
Be the first to share your thoughts!