
TL;DR — Quick Summary
Pressure
The force acting perpendicularly per unit area is called pressure. The SI unit of pressure is N/m2 or Pascal. So, Pressure is the ratio of force to surface area.
Transformation Of Pressure
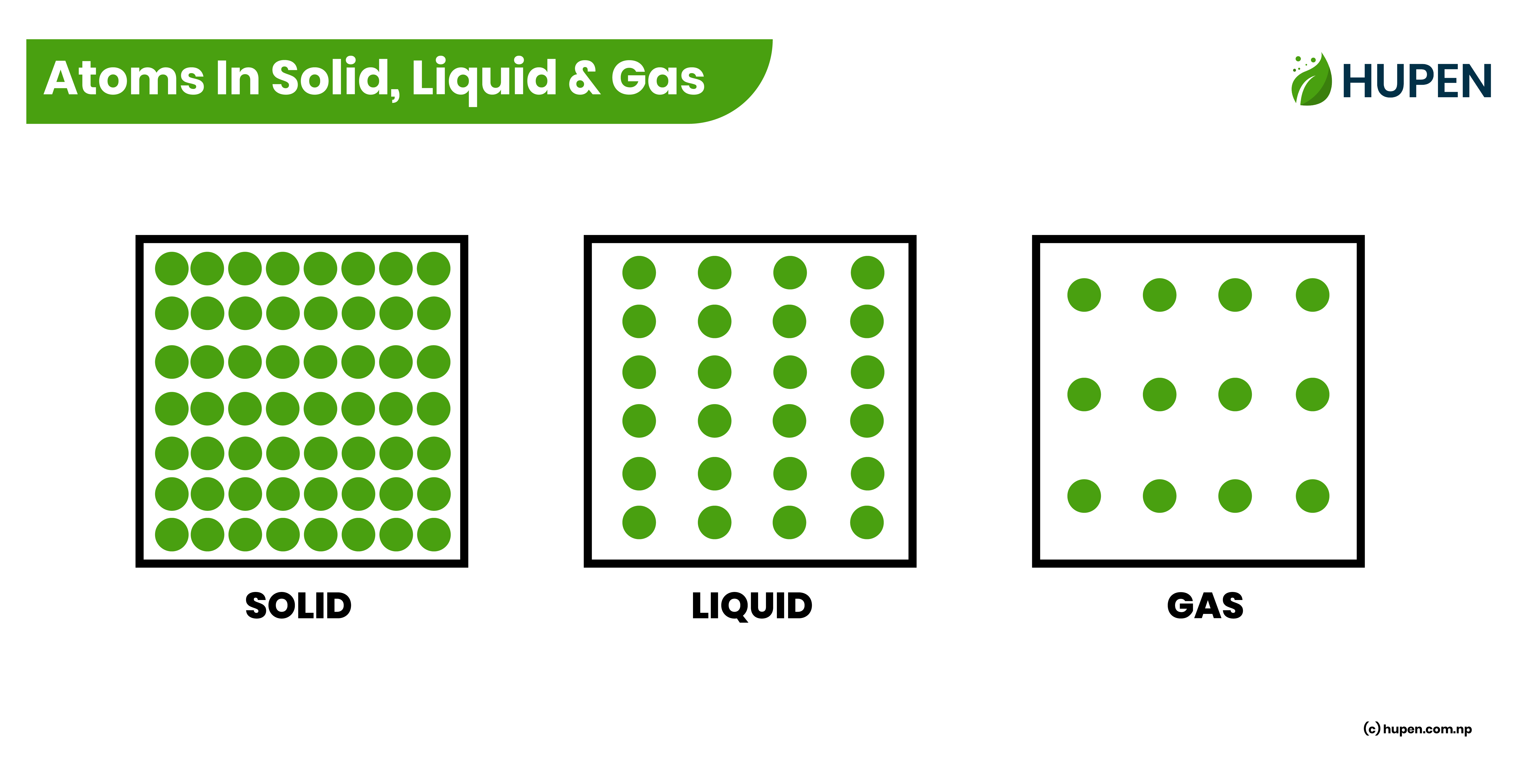
The atoms within a solid are closely bound, leaving minimal molecular space between them. As a result, the atoms are restricted in their movement, making it difficult to shift from one position to another. Consequently, pressure at a specific point within a solid does not easily propagate throughout the material. On the other hand, in fluids such as liquids and gases, the atoms can freely move between different points. This inherent mobility allows pressure to effectively transmit within the fluid when it is confined within a closed container.
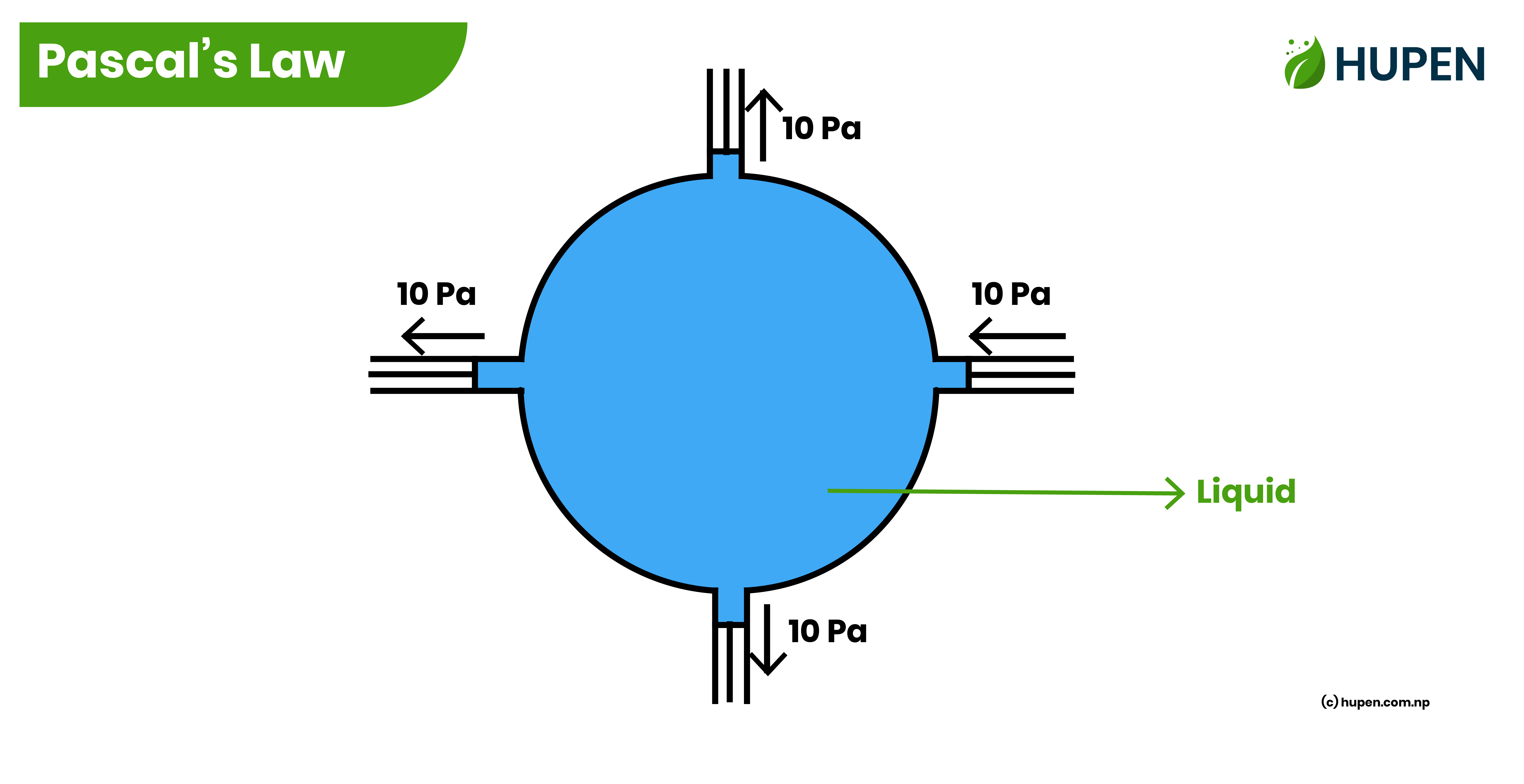
Pascal’s Law
Pascal’s law states that
“The pressure is equally transmitted perpendicularly to all sides as pressure is applied at a place on a liquid contained in a closed container.”
Applications Of Pascal’s Law
Pascal’s law is used in different Hydraulic machines like Hydraulic Press, Hydraulic Brake, Hydraulic Lift, etc.
Hydraulic Machines
The working principle of Hydraulic machines,
“A small force applied on a smaller piston is transmitted to produce a large force on the bigger piston.“
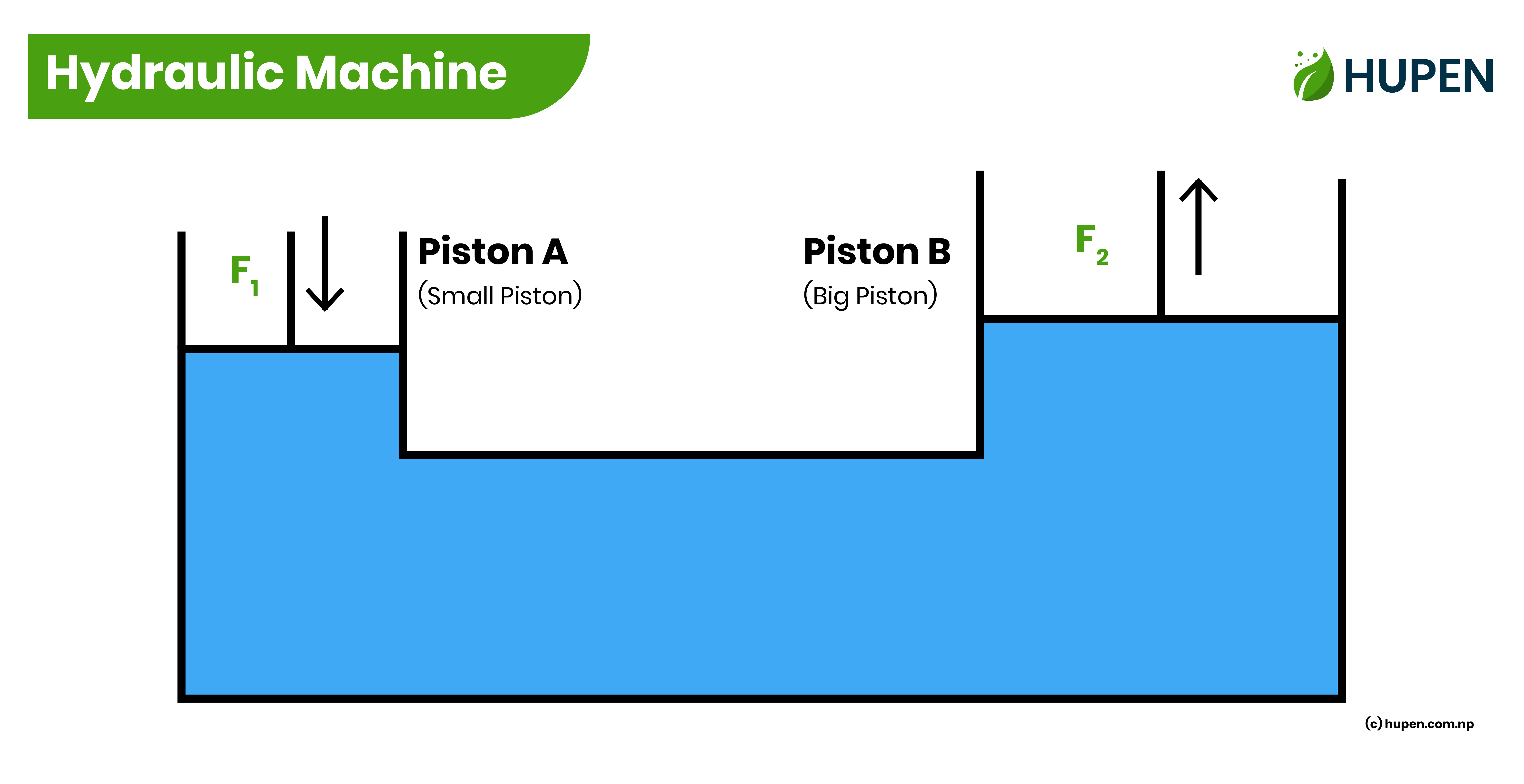
Let us consider F1 be the force on a small piston with cross-sectional area A1 and F2 be the force produced on a big piston with cross-sectional area A2.
Pressure on the small piston = Pressure on the big piston
In equation (i), the factor \((\frac{A_2}{A_1})\) is always greater than one because the cross-sectional area of the big piston is always greater than the cross-sectional area of the small piston.
Upthrust
The upward force exerted by a liquid on a body that is immersed in the liquid is known as the upthrust. It is a type of force so its SI unit is Newton (N). The main cause of upthrust is liquid pressure.
Where, W1 = Weight of an object in Air, W2 = Weight of an object in liquid
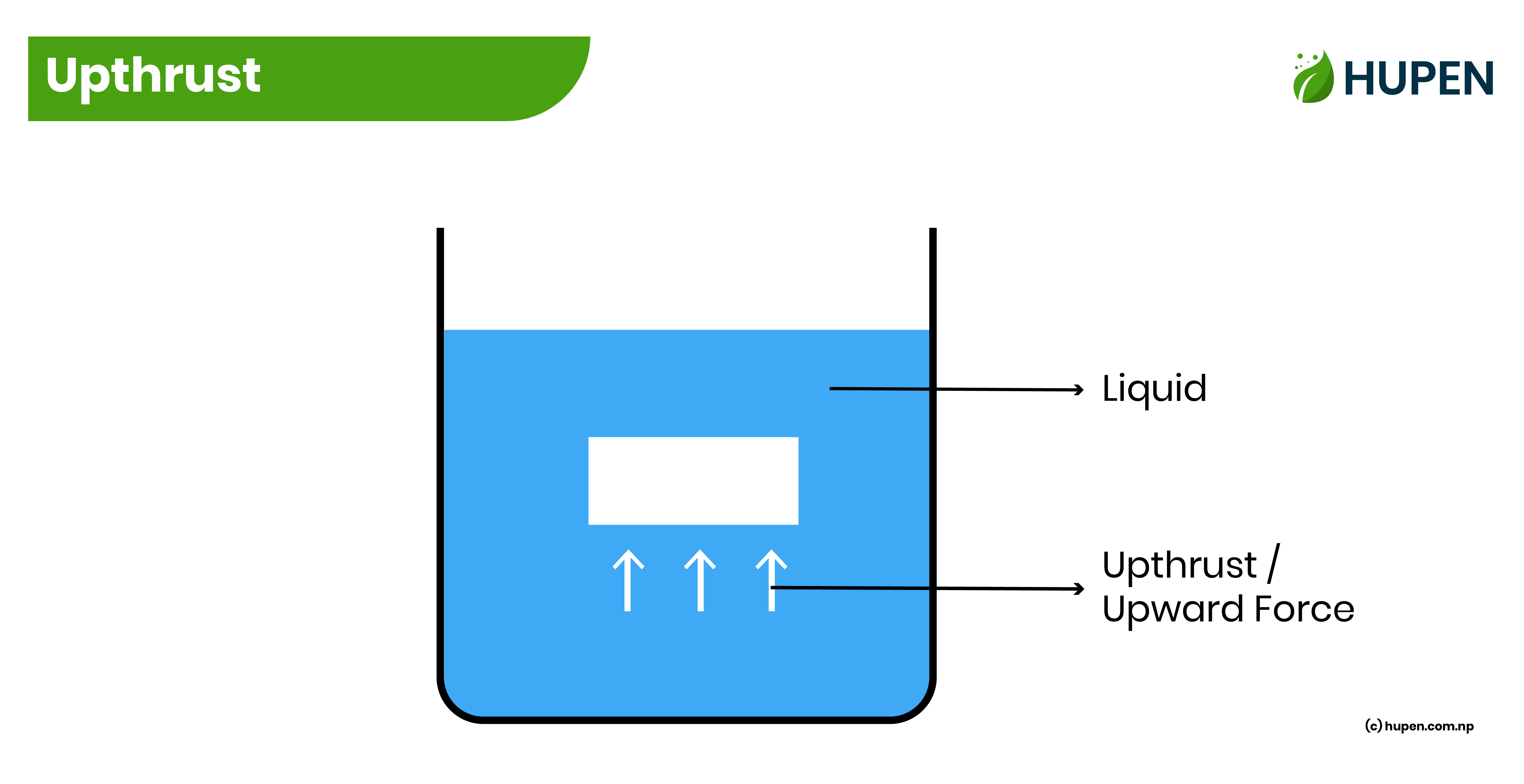
The upthrust of liquid depends on the following things.
- The density of the liquid. (\(\therefore U \propto d\))
- The volume of the displaced liquid. (\(\therefore U \propto V\))
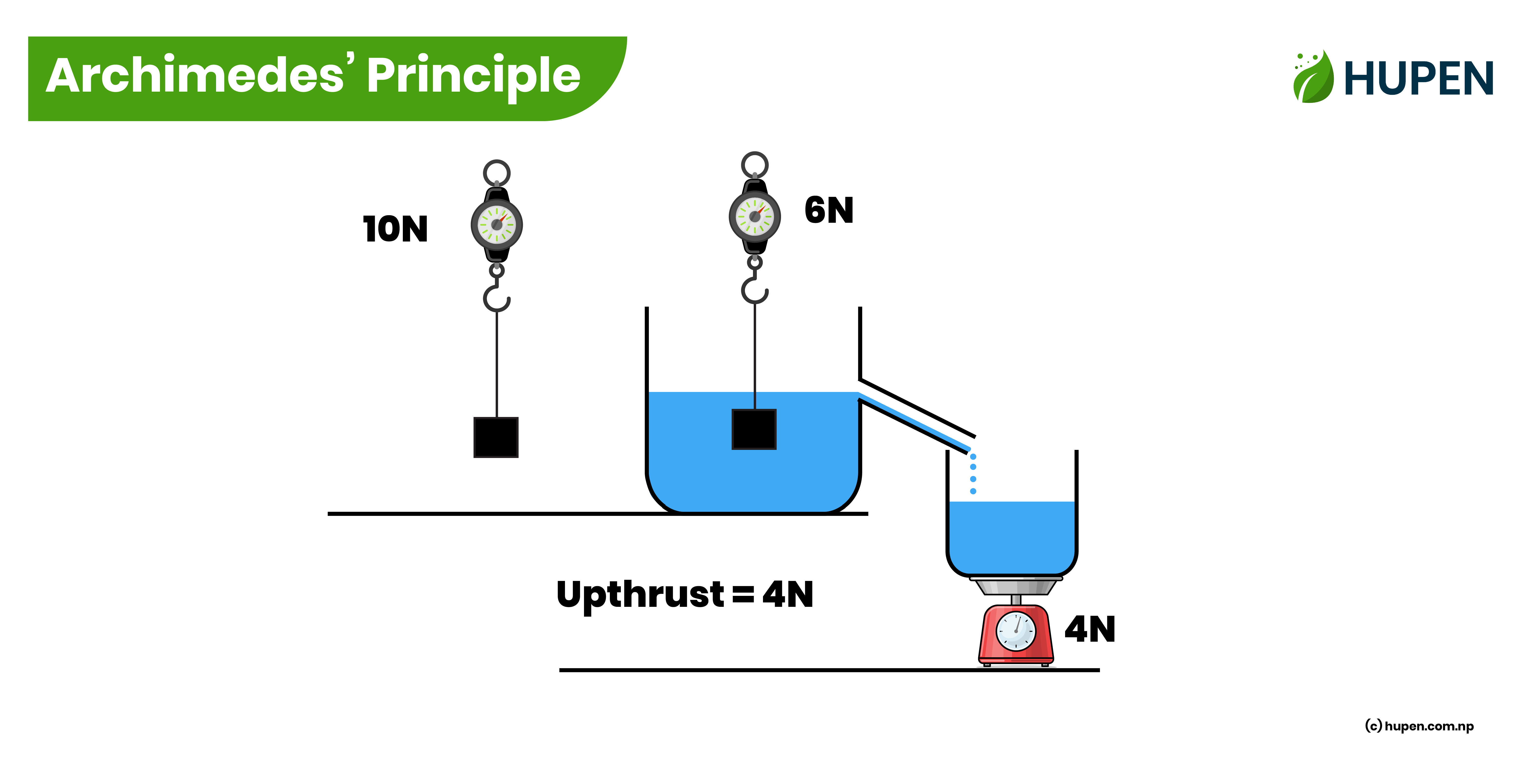
Archimedes’ Principle
The Archimedes’ Principle states that,
“When a body is partially or wholly immersed in a liquid, it experiences an upthrust which is equal to the weight of the liquid displaced by it.”
Floatation
- Floatation in Liquid
The Law of Floatation states that,
“An object floating in the liquid displaces the liquid equal to its own weight.”
- Floatation in Atmosphere
The density of the air is much less than the density of the liquid. So, the upthrust exerted by the air is also much little. Due to the upthrust exerted by the air, a hot air balloon floats in the air.
Share Now
Share to help more learners!


Chapter Discussion
0 Comments
Log in to join the discussion
No comments yet
Be the first to share your thoughts!